Action:
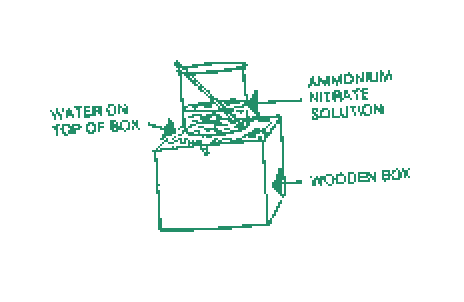
You place a white powder in a 400 ml. beaker. Put the beaker on the wet top of a small inverted wooden box. With rapid stirring, pour 100 ml. of water into the beaker. In a minute or two, you lift the beaker and the box comes along with it since it is frozen fast to the beaker.
You Need:
100 grams ammonium nitrate; small wooden box such as a chalk box; thermometer reading at least ten degrees below the freezing point of water.
Why:
A salt, which absorbs heat on dissolving, lowers the temperature of its solution below the freezing point of water.
How:
On dropping the powder into the water and stirring rapidly, heat is taken out of the solution and the temperature drops rapidly. The bottom of the beaker is cooled below the freezing point of water. The water below the beaker is frozen. This binds the beaker to the box.
Suggestions:
Do not spill any of the salt on the wet top of the box. This will prevent formation of ice where the beaker and box come in contact.
Recording the temperature at intervals makes this an interesting class project.
The beaker and ice combination can be passed around the class to show the interesting ice formation.
